|
In order to determine whether the metal acetate and ligand had formed a complex, an infrared spectroscopy (IR) and UV-vis spectroscopy was taken on both the original acetate metal and the acetate metal-ligand compound. The graphs from the IR and UV-vis were then compared in order to see if there was a significant change in appearance of the two graphs. Through this, it was determined that every experiment had formed some sort of complex. However, due to the time constraints, the structures of the complexes were not able to be determined. If more time had been available, the crystals would have been grown and sent to an X-ray crystallography lab to determine the complex structures.
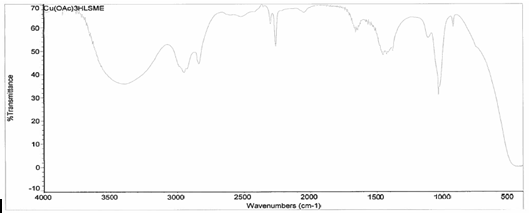
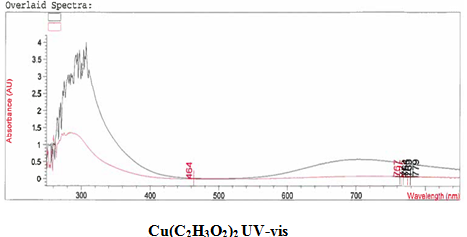
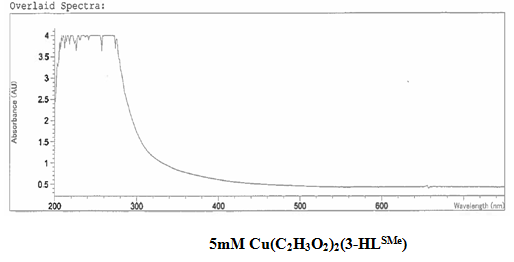
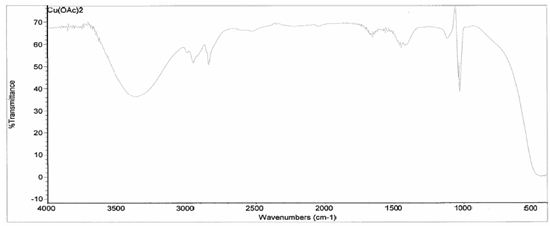
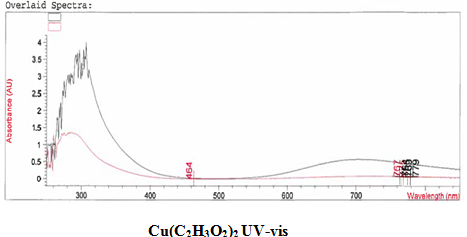
[Cu(C2H3O2)2(3-HLSMe)n]. The copper (II) acetate monohydrate and 3-HLSMe ligand solution resulted in a weight of 0.02 grams and a 86.08 percent yield. From the infrared spectroscopy (IR) taken on both the original purified copper (II) acetate monohydrate and the compound solution, it was determined that a bond had been created between the copper (II) acetate monohydrate and the 3-HLSMe ligand. This is because the compound IR has peaks in the original IR which are slightly different to that of the compound. This indicates that a change has happened within the structure. Also, in the IR of the ligand-metal compound, a new peak appeared between 2500 and 2000, which is the 3-HLSMe ligand peak. From the UV-vis taken on both the original purified copper (II) acetate monohydrate and the compound solution, it was determined that a bond had been created between the copper (II) acetate monohydrate and the 3-HLSMe ligand. This was because of the plateaus at the top of the UV-vis on the compound solution that were not on the UV-vis of the copper (II) acetate monohydrate. The wave at the end of the copper (II) acetate monohydrate UV-vis that does not appear on the compound UV-vis is not indicative of a bond between the acetate metal and the 3-HLSMe ligand. It is a result of the dark blue color of the copper atom. The reason that it shows up on the copper (II) acetate monohydrate UV-vis and not on the compound UV-vis is because they were each taken at different molarities.
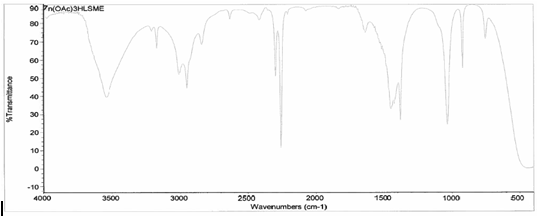
[Zn(C2H3O2)2(3-HLSMe)]. The
creation of a link between zinc (II) acetate dihydrate and the 3-HLSMe
ligand resulted in a weight of 0.035 grams and a 55.73 percent yield. From
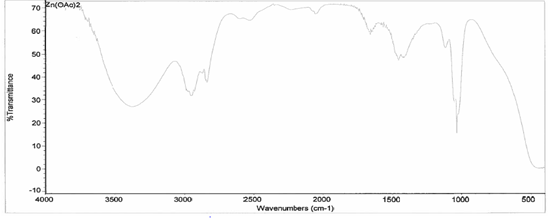
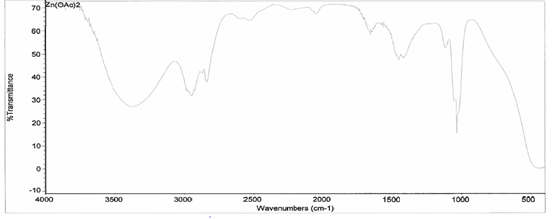
the IR taken on both the original purified zinc (II) acetate dihydrate and the
compound solution, it was determined that a bond had been created between the
zinc (II) acetate dihydrate and the 3-HLSMe ligand. This is because the
compound IR has slightly different peaks than in the original IR, which
indicates that a change has happened within the structure. Also, in the IR of
the ligand-metal compound, a new peak appeared between 2500 and 2000, which is
the 3-HLSMe ligand peak.
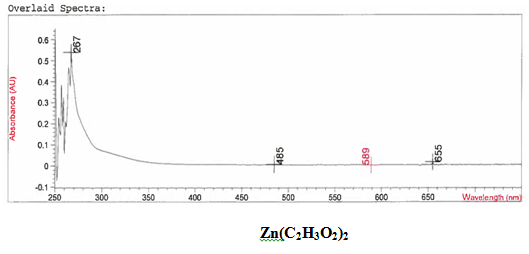
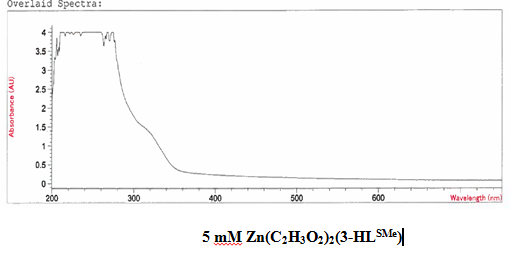
It was determined that a bond had been created between the zinc (II) acetate dihydrate and the 3-HLSMe ligand through analysis of both the UV-vis taken on the original purified zinc (II) acetate dihydrate and the compound solution. This was determined because of the plateaus at the top of the UV-vis on the compound solution that were not on the UV-vis of the zinc (II) acetate dihydrate. The peak at 310 nm that appears on the compound solution UV-vis and not on the metal acetate UV-vis is also indicative of the 3-HLSMe. The compound solution that the UV-vis was run on was very opaque, which made the details of the UV-vis on the compound harder to determine. However, the turbidity of the compound is also indicative of a ligand-acetate metal bond.
|
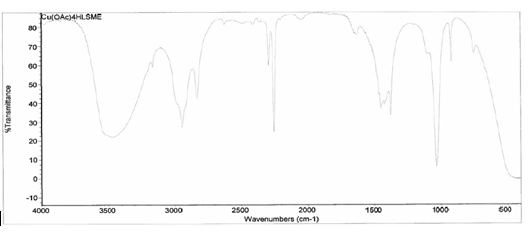
[Cu(C2H3O2)2(4-HLSMe)]. The synthesis of a link between copper (II) acetate monohydrate and the 4-HLSMe ligand resulted in a weight of 0.038 grams and a 51.34 percent yield. From the IR taken on
both the original purified copper (II) acetate monohydrate and the compound
solution, it was determined that a bond had been created between the copper
(II) acetate monohydrate and the 4-HLSMe ligand. This is because the original
copper (II) acetate monohydrate IR has slightly different peaks than the
compound solution’s IR, which indicates that a change has happened within the
structure. Also, in the IR of the ligand-metal compound a new peak appeared
between 2500 and 2000, which is the 4-HLSMe ligand peak.
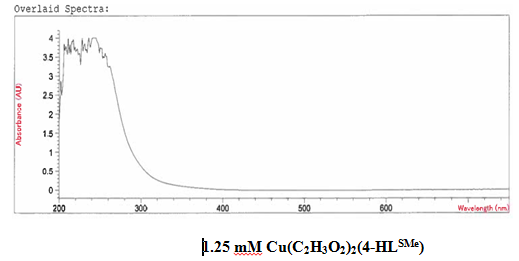
It was determined that a bond had been created between the copper (II) acetate monohydrate and the 4-HLSMe ligand through a comparison of the UV-vis taken on both the original purified copper (II) acetate monohydrate and the compound solution This was determined because of the plateaus at the top of the UV-vis on the compound solution that were not on the UV-vis of the copper (II) acetate monohydrate. The wave at the end of the copper (II) acetate monohydrate UV-vis that does not appear on the compound UV-vis is not indicative of a bond between the acetate metal and the 4-HLSMe ligand. It is a result of the dark blue color of the copper atom. The reason that it shows up on the copper (II) acetate monohydrate UV-vis and not on the compound UV-vis is because they were each taken at different molarities.
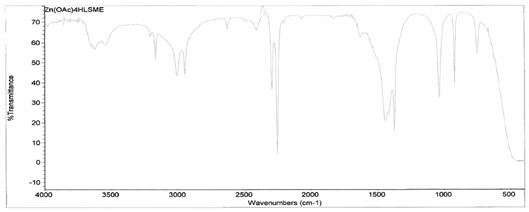
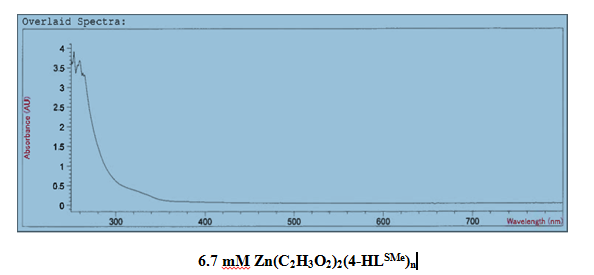
[Zn(C2H3O2)2(4-HLSMe)]. The synthesis of a link between zinc (II) acetate dihydrate and the 4-HLSMe ligand resulted in a weight of 0.04 grams and a 53.23 percent yield. From a comparison
between the IR taken on the original purified zinc (II) acetate dihydrate and
the compound solution, it was determined that a bond had been created between
the zinc (II) acetate dihydrate and the 4-HLSMe ligand. This is because the
original zinc (II) acetate dihydrate IR has slightly different peaks when
compared to the compound solution’s IR, which indicates that a change has
happened within the structure. Also, in the IR of the ligand-metal compound, a
new peak appeared between 2500 and 2000, which is the 4-HLSMe ligand peak.
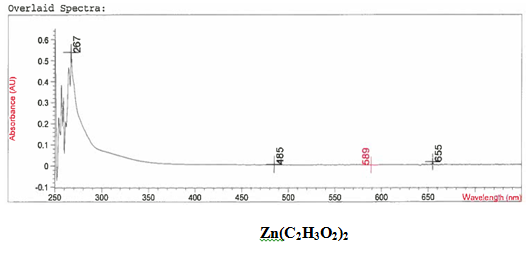
It was determined that a bond had been created between the zinc (II) acetate dihydrate and the 4-HLSMe ligand through a comparison between the UV-vis taken on the original purified zinc (II) acetate dihydrate and the UV-vis taken on the compound solution. This was determined because there is a higher absorption in the compound UV-vis than in the pure zinc (II) acetate dihydrate UV-vis.
|