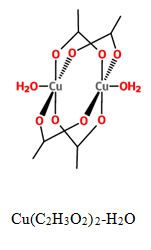
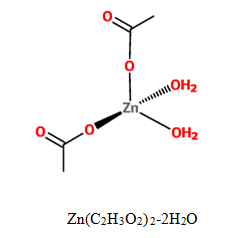
A coordination polymer is a compound
consisting of a ligand acting as a bridge between a central metal ion. Therefore, a link could be
created between transition metals through the use of ligands. However, this is
only possible if the ligand is polydentate, meaning that it would bind to one
central transition metal atom in one place and to another transition metal atom
in another.

The purpose of our study was to synthesize multiple coordination compounds with varying transition metals acting as the metal ion and polydentate 2-methylthio-N-(3-pyridinylmethyl) acetamide (3-HLSMe) ligands or polydentate 2-methylthio-N-(4-pyridinylmethyl) acetamide (4-HLSMe) ligands acting as bridges between the metal ions. Two of the transition metals being used are copper (II) acetate monohydrate (Cu(C2H3O2)2-H2O) and zinc (II) acetate dihydrate (Zn(C2H3O2)2·2H2O). By using acetates as metal nodes in the coordination complexes, new possibilities are being introduced for the architecture of supramolecular molecules. This is because a chloride (Cl-) or triflourate group (OTF-) is usually used as a counter ion and the use of an acetate as a counter ion have stronger magnetic couplings.
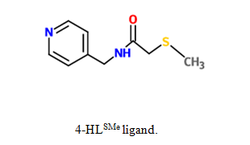
The 3-HLSMe and 4-HLSMe ligands allow multiple transition metals to be bonded together. This is due to the 3-HLSMe and 4-HLSMe ligands being polydentate. The ligand can attach to a central atom in two places, acting as a bridge between two transition metals. The ligand would attach to one transition metal atom in one place and attach also to a different metal atom in one place, creating a link between the metal atoms.
Both acetate metals used in this experiment are structured in a paddle wheel shape. Therefore, when either zinc (II) acetate dihydrate or copper (II) acetate monohydrate bonds with either the 3-HLSMe ligand or the 4-HLSMe ligand, the ligand replaces the water molecules already bonded to the metal acetate molecules. The ligand extending from the molecule would then attach to another metal acetate molecule, creating a link between the metal acetate molecules.
Both acetate metals used in this experiment are structured in a paddle wheel shape. Therefore, when either zinc (II) acetate dihydrate or copper (II) acetate monohydrate bonds with either the 3-HLSMe ligand or the 4-HLSMe ligand, the ligand replaces the water molecules already bonded to the metal acetate molecules. The ligand extending from the molecule would then attach to another metal acetate molecule, creating a link between the metal acetate molecules.